Electrodeposition Coating
Electrodeposition Coating
In electrodeposition coating, the object to be coated is dipped into the paint and electricity is applied to ensure the paint and its resin components are uniformly applied onto the surface of the object. As a result, this process makes it possible to apply coating films in circumstances where spraying and other coating methods are challenging. This process also provides a uniform film thickness.
Mechanism of the HONNYLITE® Process
The HONNYLITE process is an anodizing technique employing aluminum plus electrophoresis technology with water-soluble surface coating agents. The principle comprises the following four steps and can be performed instantaneously and continuously.
01
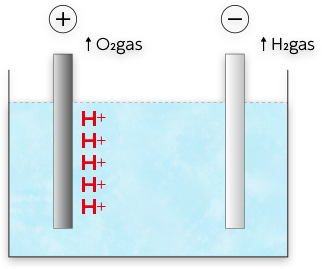
Electrolysis
Electrolysis of water generates H+ in the anode and OH– ions in the cathode.
02
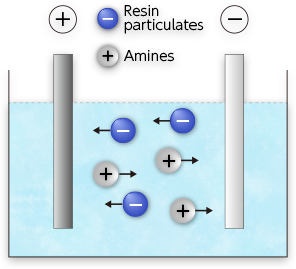
Electrophoresis
When electricity is applied, resin particles with anions move to the anode, and amines with cations move to the cathode.
03
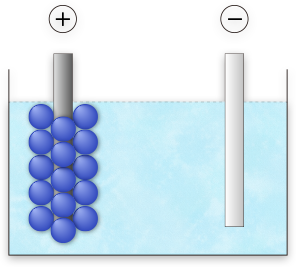
Electrodeposition
Resin particles transferred by electrophoresis collect on the anodized aluminum surface and are deposited as a surface coating agent as they lose their charge due to the neutralizing effect of H+ ions.
04
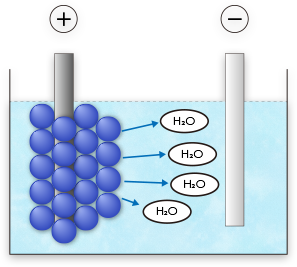
Electro-osmosis
The moisture present between the precipitated resin particles is ejected into the electrodeposition solution, and the coating film hardens firmly.
Features
Features
- Since electrodeposition takes place immediately after the aluminum anodizing process, the line processes and time required are reduced by about two-thirds.
- Because the resin film is formed electrically, it is possible to apply a uniform resin film on complex shapes and curved surfaces with excellent adhesion and corrosion-resistance characteristics.
- Sealing is performed with a water-soluble thermosetting acrylic resin film, so high-temperature baking and drying can be taken place at 170–200°C without causing cracks to form. A superb topcoat can be obtained that exhibits excellent weather resistance and chemical resistance.
Because a water-soluble resin is employed, the process is beneficial in terms of health and safety.
Painting efficiency remains quite high at 90–98%.